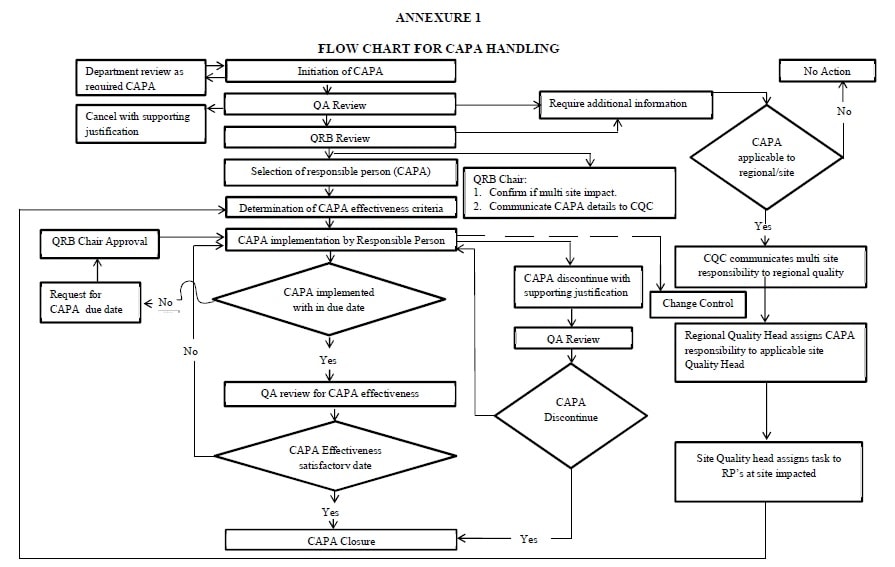
Capa processes
Corrective Action VS Preventive ActionĬorrective action is a reaction to all the causes or non-conformance that are stated above and is divided into two phases of action: On the other hand, customer grievances/suggestions, customer refusals, non-conformities raised in customer/third-party audits & recommendations by the auditors are some of the external sources that assist in finding the root cause of the problem. Through staff suggestions, document reviews, management reviews, or internal audits, it is easy to identify non-conformance internally. Note that corrective actions are mainly the actions that are based upon the identification of the problem.
#Capa processes iso#
Why Implementation of CAPA Is Necessary For QMS?Īs per FDA 21 CFR 820, ISO 9001:2015, and ISO 13485:2016, execution of corrective and preventive actions is considered as the path towards the enhancement and success of quality management systems. Records, documents, and change controlsĭuring the CAPA process, the real scope of the problem should be determined along with causes and detection methods.In the case of medical devices, CAPA is part of the different areas of the life cycle processes: It is necessary to ensure that each action taken is documented carefully because proper documentation helps in offering vital data for continuous quality improvement. At the time of the process, each step needs to be executed successfully and submissive to the CAPA program. The CAPA process consists of numerous steps. What Is the CAPA Process and Why It Is Important? The CAPA methodology needs to result in the product as well as process perfection and improved product and process understanding. To determine the root cause, an organized approach to the investigation process needs to be utilized. To guarantee that corrective and preventive actions are operative, the systematic investigation of the failure incidence is essential in recognizing the corrective and preventing actions undertaken.įor instance, a pharmaceutical or medical device company needs to have a system in place that executes corrective actions and prevents action resultant from the examination of non-conformances, product rejections, audits, deviations, governing inspections, and findings, and trends from process performance and product quality monitoring. CAPA concentrates on the methodical investigation of discrepancies so that their repetition can be avoided. In addition, it verifies the conditions and solutions for future use and then looking for other possible issues and solutions.Ĭorrective and Preventive Action is a concept inside the Good Manufacturing Practice (GMP) and ISO 13485. Steps of CAPA for Life Sciences IndustryĬAPA or corrective action and preventive action provide a structure for discovering the main cause of problems and resolving those problems.
Why Implementation of CAPA is Necessary for QMS?.What Is the CAPA Process and Why it Is Important?.We discuss how it can provide long-lasting support to your company and how digital tools could help manage the CAPA process. This article discusses the concept of corrective action and preventive action (CAPA), and the differences between the two. Therefore, various regulations and standards require an organization to take certain actions to remedy and eliminate unwanted situations.


Corrective action and preventive action (CAPA) comprises a set of actions aimed to improve an organization’s processes by eliminating the causes of recurring non-conformances and other unwanted situations.ĬAPA’s are typically used in industries like medical devices, pharmaceuticals, biotechnology, food & beverages, biologics, chemical, and others.
